Anticoagulant Use in Cutaneous Surgery
HISTORY
87-year-old man with atrial fibrillation presented with squamous cell carcinoma in situ of scalp and forehead. Patient was treated with Mohs and reconstruction on 7-3-18. Incisional hematomas appeared 2.5 weeks post Mohs and treated with I&D. Patient continued warfarin and baby aspirin throughout treatments.
DISCUSSION
Anticoagulant use in Facial and Dermatologic Surgeries has become more liberal in the last decade than it had been in the past. And that is for a good reason. Wound complications from the use of anticoagulants perioperatively is outweighed by the complications of stroke and myocardial infarctions. But what is the the real risk of the use of various anticoagulant drugs perioperatively? The most comprehensive review of facial plastic surgical cases at University of Michigan (2015) found some interesting observations.
- Warfarin – 4x bleeding, 7x post-op infection, no increase in serious complications (flap necrosis, dehiscence, return to OR)
- Aspirin – not associated with increased complications in cutaneous surgeries (increased in body cavity surgeries)
- Clopidogrel – no increase in bleeding in cutaneous surgeries
- Multiple anticoagulant meds – increase in complications
So, Aspirin and Clopidogrel (Plavix) do not increase the risk of bleeding in cutaneous and facial surgeries. While, warfarin and the use of multiple anticoagulants increase the risk.
The newer direct oral anticoagulants (DOAC’s) such as Pradaxa (dabigatran), Xarelto (rivaroxaban), and Eliquis (apixaban), and Lixiana (edoxoban) offer the advantage of a short half-life. This allows brief termination of their use, thus minimizing thromboembolic complications. A summary article published in 2018 in Research and Practice in Thrombosis and Haemostasis found that interruption of DOAC’s in atrial fibrillation resulted in 0.4% risk of thromboembolic events. Continued use of DOAC’s perioperatively caused 1.8% risk of major bleeding. The article references a protocol for interruption of these drugs.
For low bleeding risk procedures, which would encompass most cutaneous and facial plastic surgeries, stopping DOAC’s one day before and the day of the operation is considered an acceptable risk. For minimal bleeding risk procedures, continuation of these drugs perioperatively is recommended. Pradaxa (dabigatran) recommendations are different for patients with decreased eGFR. For high bleeding risk procedures, interruption begins two days pre-op and the anticoagulant is restarted post-op day three. This protocol is currently under study to validate these recommendations.
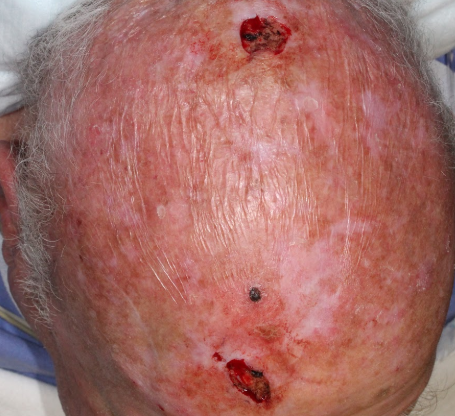
Fig. 1. Mohs excision defects.
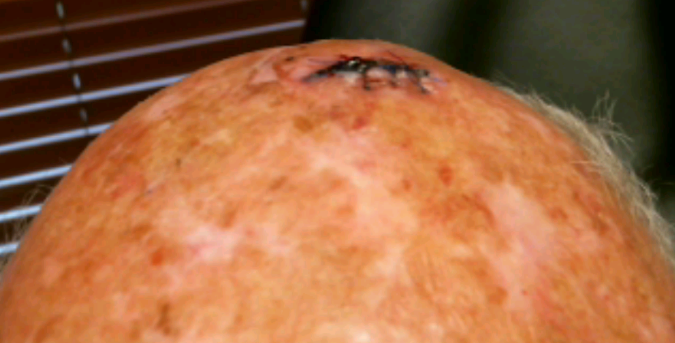
Fig. 2. Incisional hematoma 2.5 weeks post closure.
